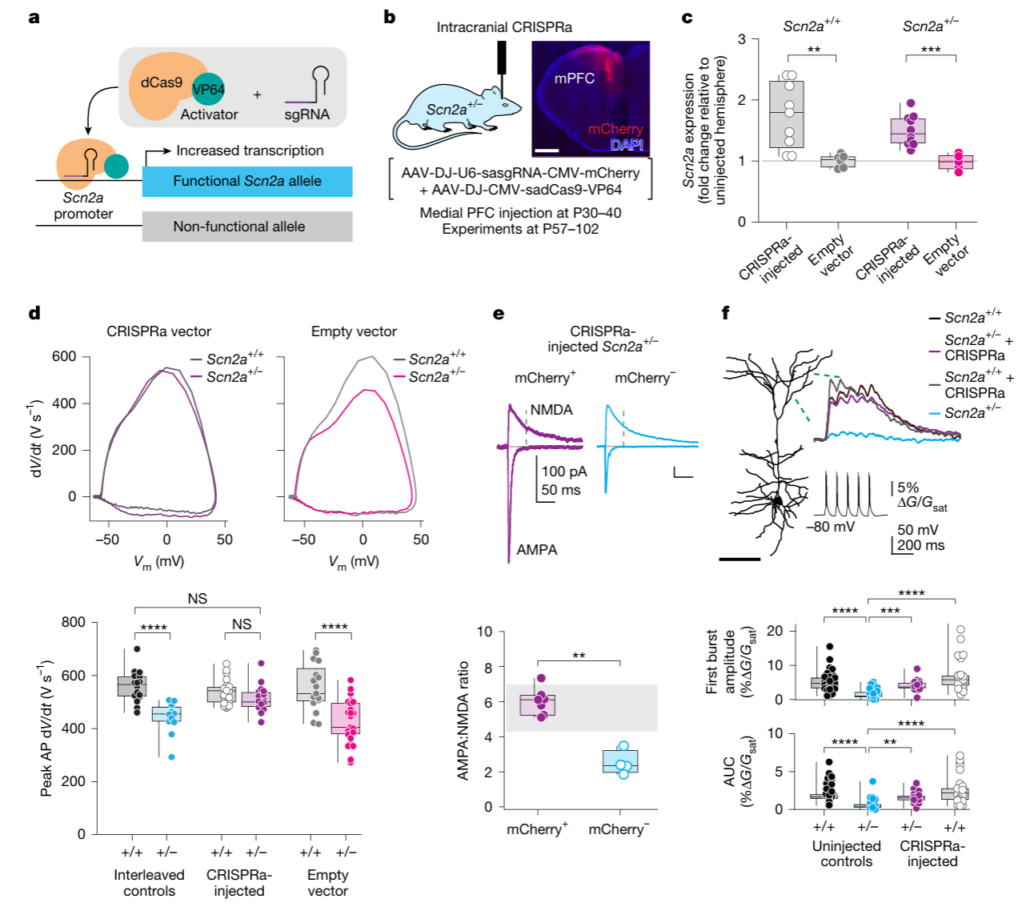
Former Jenkins lab PhD student, Andrew Nelson, along with co-first authors Serena Tamura and Perry Spratt have published their magnum opus on the use of CRISPR-activation (CRISPRa) to correct Scn2a haploinsufficiency in the journal Nature. Led by the Kevin Bender and Nadav Ahituv labs at UCSF, this study uses a promoter-targeted dCas9 fusion protein to upregulate the native Scn2a allele in heterozygous loss-of-function mice. Our collaborative team showed that CRISPRa can upregulate Scn2a/Nav1.2, even in adult animals, to rescue cellular, synaptic, and behavioral deficits, laying the groundwork for potential future gene therapy approaches for patients with SCN2A loss-of-function variants. Interestingly, Scn2a-directed CRISPRa in wild-type mice did not “overshoot” normal Nav1.2 function of promote seizure susceptibility, suggesting that there is a “ceiling effect” promoting safety. This work joins the growing number of studies showing the exciting potential of sodium channel gene therapies in rescuing severe disease, like the work of Dr. Lori Isom and Stoke Therapeutics in children carrying SCN1A variants in Dravet Syndrome. Read more about our collaborative study here. Congrats to the whole team!!